A vascularized, innervated, 3D skin-on-a-chip organoid model to study viral infections and antiviral treatment
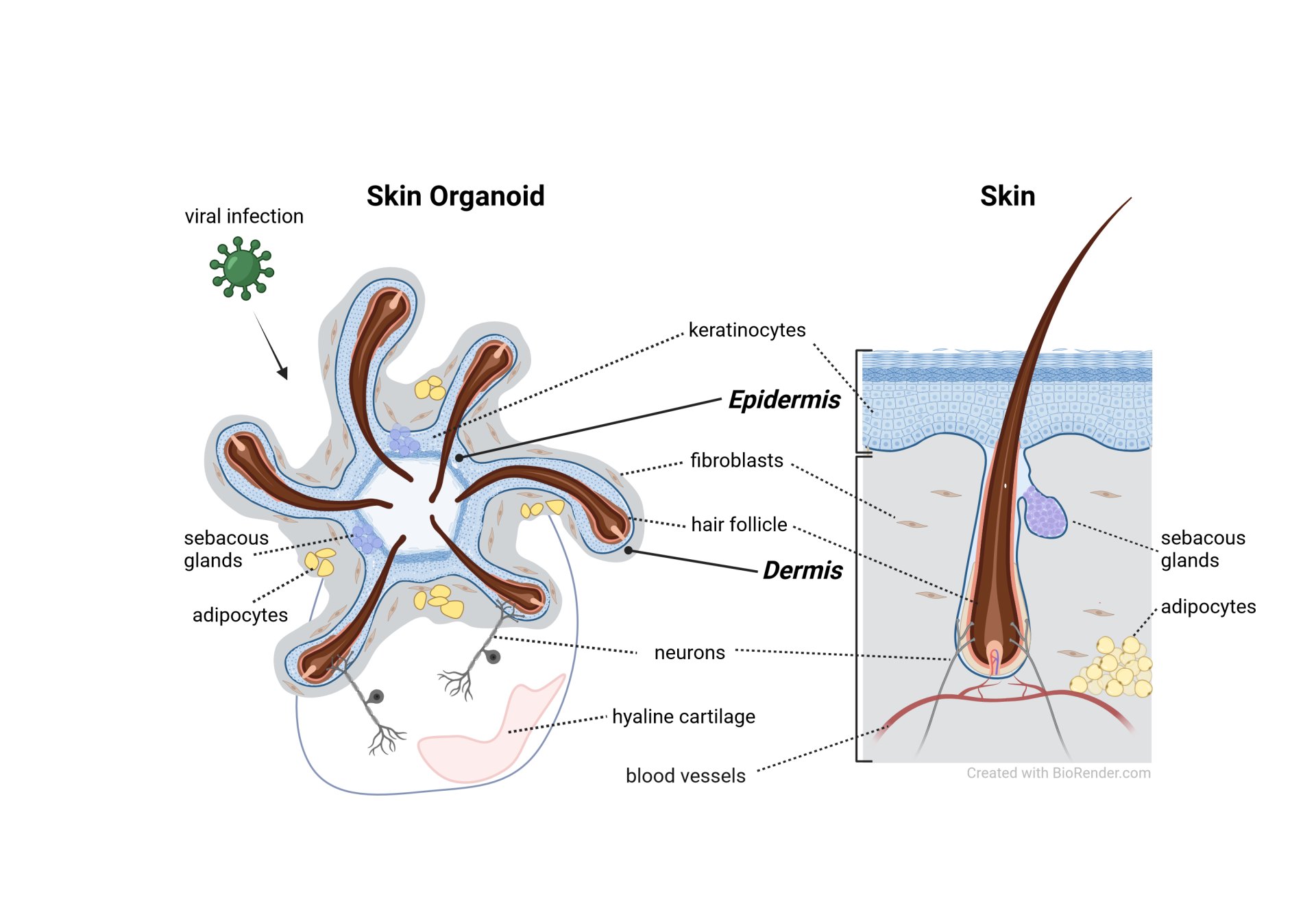
Physiological model systems and advanced analysis tools are the basis for a comprehensive understanding of infectious diseases and development of therapeutic strategies. Skin is a complex multifunctional organ that serves as a first barrier against pathogens but is also a site of entry, pathogenesis, and persistence of pathogens including viruses. Traditional in vitro models fail to reproduce the distinct structural and functional features of human skin, which hinders their ability to provide an authentic assessment of the life cycle and pathogenesis of viruses that target the skin. Following a recently published protocol (Lee et al. Nat Protoc. 2022), we established a highly complex human skin organoid model derived from human induced pluripotent stem cells. It contains an epidermis, a dermis with adipose tissue, pigmented hair-producing hair follicles, sebaceous glands, and a complex innervation system. Despite advanced organoid differentiation protocols, certain applications and aspects of organoid systems remain demanding. These challenges include the absence of immune cells, vascularization, and their utilization as infection models. In response to these limitations, this interdisciplinary project combines expertise on viral infections and organoids, microfluidic chips, and 3D high-resolution X-ray based microscopy to create a vascularized, innervated, 3D skin-on-a-chip organoid model for next-level research on human pathogenic skin tropic viral infections. This innovative approach will improve the practicality and physiological relevance not only for the skin organoid system but also for other advanced in vitro systems and their use in infectious diseases research.




